UPDATE: FDA Advisers Unanimously Recommend Moderna’s Covid-19 Vaccine for Ages 6-17 Despite Data Showing High Risks of Heart Inflammation
UPDATE: FDA Advisers Unanimously Recommend Moderna’s Covid-19 Vaccine for Ages 6-17 Despite Data Showing High Risks of Heart Inflammation
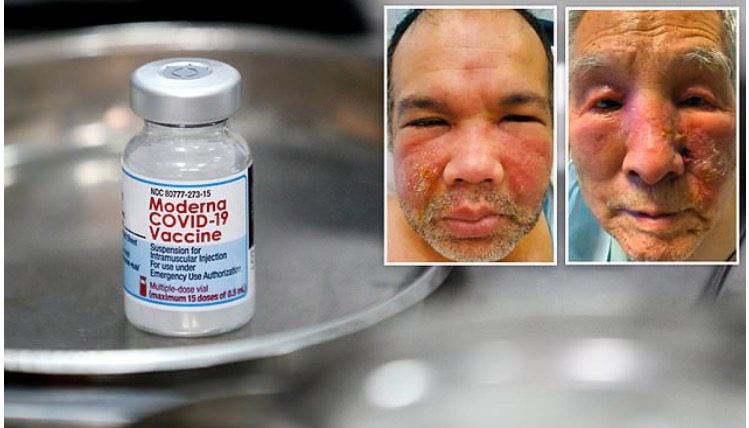
Moderna’s COVID-19 vaccine may have a higher risk of heart inflammation than the Pfizer shot based on recent data, the U.S. Centers for Disease Control and Prevention said on Tuesday.
On Tuesday, the U.S. Food and Drug Administration met with its Vaccines and Related Biological Products Advisory Committee (VRBPAC) to discuss Moderna’s EUA request for a COVID-19 vaccine for children 6 through 17 years of age.
The same expert panel will meet again on Wednesday to discuss shots from Moderna and Pfizer for the kids under 5.
According to the CDC data, the incidence of heart inflammation was 4.41 excess per 100,000 who received the Pfizer/BioNTech for males aged 18-39 versus 6.27 excess cases per 100,000 for Moderna, Reuters reported.
Daniel Horowitz, the senior editor at The Blaze, tweeted on Monday regarding the side effects of the Moderna vaccine.
“Holy Moly! one-quarter of the kids in Moderna’s trial reported Grade 3 side effects, meaning they couldn’t go to school fda.gov/media/159189/d So many had flu-like symptoms! Even the original strain of covid mainly did this to kids. Yet, we are giving them these symptoms upfront,” Horowitz tweeted.
Holy Moly! one quarter of the kids in Moderna’s trial reported GRade 3 side effects, meaning they couldnt go to school https://t.co/93eO3zdd3E So many had flu like symptoms! Even the original strain of covid mainly did this to kids.Yet, we are giving them these symptoms up front pic.twitter.com/j9uAjXqd6I
— Daniel Horowitz (@RMConservative) June 14, 2022
The FDA called on speakers about their views on authorizing the Moderna vaccine for children ages 6 to 17. Some of the invited speakers were not in favor of the vaccine due to its life-threatening risks that completely ruined their lives.
Robyn Handsman was one of the speakers invited to share her first-hand experience with the experimental Moderna vaccine. Mrs. Handsman got her first Moderna shot on September 15, 2021. The following day, she woke up and her hands and arms were completely numb and tingly. Her blood pressure was also extremely high. After five days, her doctor informed her that she has protein in her urine. She was diagnosed also with permanent kidney damage.
“Why on Earth would you give this to kids? And young people when the risk of dying from COVID is practically zero. How many people have to go to sleep and never wake up? It is not a coincidence. Kids are dying on the fields from heart conditions like never before. Me and thousands and thousands of lives are forever changed. We have no recourse. We can’t go after the vaccine injury program. Moderna has complete immunity and we’re censored on our support groups. We have to talk in code, and I’m very concerned that this is all about money and I really don’t know how people sleep at night knowing this is injuring and killing people all over the place,” Handsman said.
Dr. Harvey Klein also gave his honest and brutal comments regarding the vaccine. Dr. Harvey A. Klein is an orthopedist in Brooklyn, New York and is affiliated with multiple hospitals.
“I’m appalled at the arrogance that you meaning the FDA [has] in even thinking about vaccinating healthy children with outdated, highly toxic, Covid vaccines. Children who have a 99.98% survival rate. Why in the world would you want to try to improve on perfection by exposing them to significant chances of being permanently severed, severely injured, or dead? The risk is infinite and the benefits are nonexistent, and the efficacy is extremely negative. Why do you want to mess with God’s given perfect system? The best treatment you can do is to leave these children alone under God’s care and failing to do that, then your only purpose is to name and kill. That clearly being the case, the FDA should change its name to the JMI, the Josef Mengele Institute,” Klein explained.
After the committee meeting, the FDA advisory panel unanimously recommended Moderna’s COVID-19 vaccine for emergency use authorization for children aged 6 to 17, saying it would offer more benefits than risks.
The following questions were asked of the 22 voting members for today’s meeting.
1. Based on the totality of scientific evidence available, do the benefits of the Moderna COVID-19 Vaccine when administered as a 2-dose series (100 ug each dose) outweigh its risks for use in adolescents 12 through 17 years of age?
- 22 members unanimously believed Moderna Covid vaccine outweigh its risks for use in adolescents 12 through 17 years of age
2. Based on the totality of scientific evidence available, do the benefits of the Moderna COVID-19 Vaccine when administered as a 2-dose series (50 ug each dose) outweigh its risks for use in children 6 through 11 years of age?
- 22 members unanimously believed Moderna Covid vaccine outweigh its risks for use in children 6 through 11 years of age
“The FDA, which usually follows the advice of the committee, is likely to give final approval to Moderna’s vaccine in this age group soon,” FOX reported.
The post UPDATE: FDA Advisers Unanimously Recommend Moderna’s Covid-19 Vaccine for Ages 6-17 Despite Data Showing High Risks of Heart Inflammation appeared first on The Gateway Pundit.
Go to Source
Author: Jim Hoft